
The IT company itself has to consistently implement the agreed quality assurance measures and thus moves into the focus of the regulatory authorities. If an organization in the regulated context wants to empower itself with works of an IT supplier, then the approach of initial verification of adequateness and reliability of the supplier also applies here.
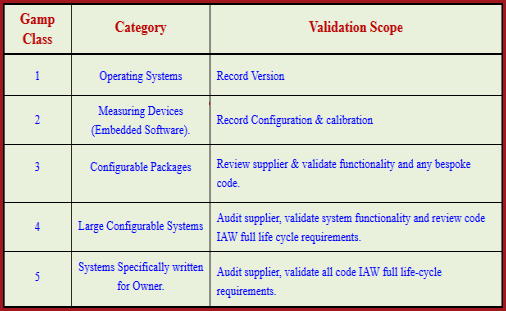
The resulting monitoring of the entire lifecycle allows the optimization of procedures and processes, such as the avoidance of duplicate activities through the integration of computer system activities. These must then be implemented and verified accordingly. Based on these functions, the functional risk assessment begins and the scope and number of checks for the computer-based system are specified. Afterwards, the functions are determined which may affect the data security, the product quality and the data integrity. Initially, a basic risk evaluation is conducted and then the effects of the system are analyzed. These affect the perspectives and procedures and deliberate decisions must be made. The validation efforts according to GAMP in practise depend on the parameters for data security, the complexity and novelty of the IT system used. However, the GAMP5 is not a standard and therefore has no binding character. Thus it has evolved as the standard set of rules for the validation of computer-based systems in the pharmaceutical industry. GAMP5 is named as an industry standard as it is also referred to in official documents. These are always risk-based and summarize many other guidelines and standards in the pharmaceutical industry. It includes various and scalable calidation approaches and strategies. GAMP5 offers a pragmatic and practical guide for the implementation of a GxP-compliant conputer-based system. The number five represents version 5 which is currently the most up-to-date implementation. GAMP stands for Good Automated Manufacturing Practice.
